| Micrococcus luteus |
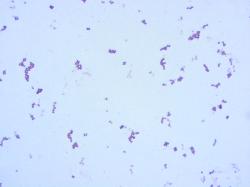
| Micrococcus luteus, Gram-stained cells |
| Micrococcus luteus yellow-pigmented colonies on Mueller-Hinton agar |
Taxonomy
Morphology
Cultural characteristics
Biochemical characters
Ecology
Pathogenicity
References
Phylum Actinomycetota, Class Actinomycetes, Order Micrococcales, Family Micrococcaceae, Genus Micrococcus, Micrococcus luteus
(Schroeter 1872) Cohn 1872, emend. Wieser et al., 2002, type species of the genus.
Three biovars were proposed by Wieser et al. (2002).
Old synonyms: Bacteridium luteum Schroeter 1872, Sarcina lutea (Schroeter 1872) Schroeter 1886, Micrococcus lysodeikticus
Fleming 1933, "not Micrococcus luteus" Lehmann and Neumann 1896.
Note: Tang and Gillevet, propose to reclassify the ATCC 9341 strain as Kocuria
rhizophila. This strain was previously designated as a quality-control strain in a
number of papers and it is also cited as the standard culture in several official
methods and manuals.
(Schroeter 1872) Cohn 1872, emend. Wieser et al., 2002, type species of the genus.
Three biovars were proposed by Wieser et al. (2002).
Old synonyms: Bacteridium luteum Schroeter 1872, Sarcina lutea (Schroeter 1872) Schroeter 1886, Micrococcus lysodeikticus
Fleming 1933, "not Micrococcus luteus" Lehmann and Neumann 1896.
Note: Tang and Gillevet, propose to reclassify the ATCC 9341 strain as Kocuria
rhizophila. This strain was previously designated as a quality-control strain in a
number of papers and it is also cited as the standard culture in several official
methods and manuals.
Gram-positive cocci, occuring in tetrads and in irregular clusters of tetrads. Nonmotile.
Endospores are not formed.
Endospores are not formed.
Colonies are smooth, convex with regular edge, yellow, yellowish green, or orange
pigmented, approximately 4 mm after 2-3 days on PYES-agar. Some strains form a
violet pigment which diffuses into the medium. Colonies forming cubical packets may
have a granular surface and a matt appearance. Optimal growth at 25-37 ºC. Can
grow at 15 ºC. Growth or weak growth is observed at 45 ºC, at pH 10.0, and in the
presence of 10% NaCl. No growth in the presence of 15% NaCl or at 4 ºC. Aerobic.
Grows on common media containing yeast extract and peptone and a pH close to 7.0.
Biovar 1 does not grow at pH 6.
pigmented, approximately 4 mm after 2-3 days on PYES-agar. Some strains form a
violet pigment which diffuses into the medium. Colonies forming cubical packets may
have a granular surface and a matt appearance. Optimal growth at 25-37 ºC. Can
grow at 15 ºC. Growth or weak growth is observed at 45 ºC, at pH 10.0, and in the
presence of 10% NaCl. No growth in the presence of 15% NaCl or at 4 ºC. Aerobic.
Grows on common media containing yeast extract and peptone and a pH close to 7.0.
Biovar 1 does not grow at pH 6.
Isolated from human and animal skin, clinical samples and foods.
Biovar 2 strains were isolated from a medieval wall painting and from indoor air.
Biovar 3 strain was isolated from an activated-sludge plant in Ballarat, Australia.
Resistant to furazolidone (0.03% w/v). Susceptible to penicillin, erythromycin,
streptomycin, methicillin, novobiocin, tetracycline, chloramphenicol, neomycin and
polymyxin B. Cell walls are susceptible to lysozyme.
Biovar 2 strains were isolated from a medieval wall painting and from indoor air.
Biovar 3 strain was isolated from an activated-sludge plant in Ballarat, Australia.
Resistant to furazolidone (0.03% w/v). Susceptible to penicillin, erythromycin,
streptomycin, methicillin, novobiocin, tetracycline, chloramphenicol, neomycin and
polymyxin B. Cell walls are susceptible to lysozyme.
Opportunistic pathogen. May produce septic shock, meningitis, septic arthritis,
endocarditis, infections associated with indwelling lines, continuous ambulatory
peritoneal dialysis, or a ventriculo-peritoneal shunt, intracranial suppuration,
bacteremia, chronic cutaneous infections in HIV-positive patients, and catheter
infection.
Can poduce cadaverine in food through the action of lysine decarboxylase. Cadaverine
has little toxicity but it potentiates the toxicity of histamine.
endocarditis, infections associated with indwelling lines, continuous ambulatory
peritoneal dialysis, or a ventriculo-peritoneal shunt, intracranial suppuration,
bacteremia, chronic cutaneous infections in HIV-positive patients, and catheter
infection.
Can poduce cadaverine in food through the action of lysine decarboxylase. Cadaverine
has little toxicity but it potentiates the toxicity of histamine.
- Hans-Jurgen Busse, 2012. Family I. Micrococcaceae Pribham 1929, 361 AL emend. Stackebrandt, Rainey and Ward-Rainey 1997,
479 in: Bergey’s Manual of Systematic Bacteriology, second edition, Volume Five The Actinobacteria, Part A, Springer, 571-666. - Wieaser M., Denner E.B.M., Kampfer P., Schumann P., Tindall B., Steiner U., Vybiral D., Lubitz W., Maszenan A.M., Patel B.K.C.,
Seviour R.J., Radax C. and Busse H.J.: Emended descriptions of the genus Micrococcus, Micrococcus luteus (Cohn 1872) and
Micrococcus lylae (Kloos et al. 1974). Int. J. Syst. Evol. Microbiol., 2002, 52, 629-637. - Liu H., Xu Y., Ma Y. and Zhou P.: Characterization of Micrococcus antarcticus sp. nov., a psychrophilic bacterium from Antarctica. Int.
J. Syst. Evol. Microbiol., 2000, 50, 715-719. - Chen H.H., Zhao G.Z., Park D.J., Zhang Y.Q., Xu L.H., Lee J.C., Kim C.J. and Li W.J.: Micrococcus endophyticus sp. nov., isolated
from surface-sterilized Aquilaria sinensis roots. Int. J. Syst. Evol. Microbiol., 2009, 59, 1070-1075. - Rakash, O., Nimonkar, Y., Munot, H., Sharma, A., Vemuluri, V. R., Chavadar, M. S. and Shouche, Y. S. 2014. Description of
Micrococcus aloeverae sp. nov., an endophytic actinobacterium isolated from Aloe vera. Int. J. Syst. Evol. Microbiol., 64, 3427-3433. - Salton, M. R. J. 1958. The Lysis of Micro-organisms by Lysozyme and Related Enzymes. J. gen. Microbiol. 18, 481-490.
- Tang J.S. and Gillevet P.M.: Reclassification of ATCC 9341from Micrococcus luteus to Kocuria rhizophila. Int. J. Syst. Evol. Microbiol.,
2003, 53, 995-997. - Goodfellow, M., T.M. Embley and B. Austin. 1985. Numerical taxonomy and emended description of Renibacterium salmoninarum.
J. Gen. Microbiol. 131: 2739-2752.
Positive results for acid and alkaline phosphatase, chymotrypsinase, catalase,
esterase C8, alpha-glucosidase, methyl red test, oxidase, acid production from
glycerol, D-arabinose, arbutin, fructose, glucose (contradictory results among
authors), methyl alpha-D-glucoside, mannose, maltose, sucrose, salicin and
trehalose.
Can assimilate D-glucose, maltose, D-mannose, pyruvate, propionate, sucrose and
D-trehalose.
Negative results for arginine dihydrolase, cystine arylamidase, esterase C4, esculin
hydrolysis, alpha- and beta-glactosidase, beta-glucuronidase, beta-glucosidase,
alpha-mannosidase, nitrate reduction, starch hydrolysis, trypsinase, valine
arylamidase, Voges-Proskauer test, acid production from adonitol, L-arabinose,
cellobiose, dulcitol, erythritol, esculin, D-fucose, galactose, beta-gentiobiose,
glycogen, gluconate, inositol, inulin, lactose, D-lyxose, mannitol, melibiose,
melezitose, methyl alpha-D-mannoside, N-acetylglucosamine, raffinose, rhamnose,
ribose, starch, sorbitol, sorbose, D-tagatose, xylitol, L- and D-xylose.
No assimilation of D-fructose, N-acetyl-D-glucosamine, L-rhamnose, gluconate,
cis-aconitate, trans-aconitate, adipate, azelate, itaconate, L-malate, mesaconate,
suberate, beta-alanine, L-ornithine, L-tryptophan, L-leucine, 3-hydroxybenzoate, and
4-hydroxybenzoate.
Variable results for Tween 80 hydrolysis, urease, utilization of acetate and L-alanine.
esterase C8, alpha-glucosidase, methyl red test, oxidase, acid production from
glycerol, D-arabinose, arbutin, fructose, glucose (contradictory results among
authors), methyl alpha-D-glucoside, mannose, maltose, sucrose, salicin and
trehalose.
Can assimilate D-glucose, maltose, D-mannose, pyruvate, propionate, sucrose and
D-trehalose.
Negative results for arginine dihydrolase, cystine arylamidase, esterase C4, esculin
hydrolysis, alpha- and beta-glactosidase, beta-glucuronidase, beta-glucosidase,
alpha-mannosidase, nitrate reduction, starch hydrolysis, trypsinase, valine
arylamidase, Voges-Proskauer test, acid production from adonitol, L-arabinose,
cellobiose, dulcitol, erythritol, esculin, D-fucose, galactose, beta-gentiobiose,
glycogen, gluconate, inositol, inulin, lactose, D-lyxose, mannitol, melibiose,
melezitose, methyl alpha-D-mannoside, N-acetylglucosamine, raffinose, rhamnose,
ribose, starch, sorbitol, sorbose, D-tagatose, xylitol, L- and D-xylose.
No assimilation of D-fructose, N-acetyl-D-glucosamine, L-rhamnose, gluconate,
cis-aconitate, trans-aconitate, adipate, azelate, itaconate, L-malate, mesaconate,
suberate, beta-alanine, L-ornithine, L-tryptophan, L-leucine, 3-hydroxybenzoate, and
4-hydroxybenzoate.
Variable results for Tween 80 hydrolysis, urease, utilization of acetate and L-alanine.
(c) Costin Stoica

| Antibiogram |
| Encyclopedia |
| Culture media |
| Biochemical tests |
| Stainings |
| Images |
| Movies |
| Articles |
| Identification |
| Software |
| R E G N U M PROKARYOTAE |

Differential characters of the Micrococcus luteus biovars (assimilation tests):
|
|
|


| Back |