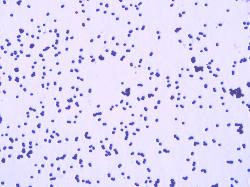
| K. rosea Gram-stained cells |
| Kocuria rosea salmon-red pigment production on Mueller Hinton agar |
| Kocuria rosea |
Taxonomy
Morphology
Cultural characteristics
Biochemical characters
Ecology
Pathogenicity
References
Phylum Actinomycetota, Class Actinomycetes, Order Micrococcales, Family Micrococcaceae, Genus Kocuria, Kocuria rosea (Flugge
1886) Stackebrandt et al. 1995, type species of the genus.
Basonym: Micrococcus roseus Flugge 1886.
The species Kocuria rosea and Kocuria erythromyxa are united, Kocuria rosea having priority over Kocuria erythromyxa.
Old synonyms: Sarcina erythromyxa Chester 1901, Deinococcus erythromyxa (ex
Chester 1901) Brooks and Murray 1981.
1886) Stackebrandt et al. 1995, type species of the genus.
Basonym: Micrococcus roseus Flugge 1886.
The species Kocuria rosea and Kocuria erythromyxa are united, Kocuria rosea having priority over Kocuria erythromyxa.
Old synonyms: Sarcina erythromyxa Chester 1901, Deinococcus erythromyxa (ex
Chester 1901) Brooks and Murray 1981.
Gram-positive cocci, 1-1.5 μm in diameter, occuring in pairs, tetrads and clusters.
Endospores are not formed. Nonmotile. Nonencapsulated.
Endospores are not formed. Nonmotile. Nonencapsulated.
Colonies are circular, slightly convex, smooth (occasionally rough), and pink or red,
becoming more distinct with age. No water-soluble exopigment is formed. Growth is
stimulated by cysteine and thiamine or cysteine, thiamine, and pantothenic acid. In
nutrient broth produce moderate turbidity and deposit; in some strains a surface
ring and pellicle are formed. The optimal growth temperature is in the range 25-37 ºC.
No growth at 5 ºC. Grows well in 7.5% NaCl media. No growth in 10 or 15% NaCl
media. Aerobic. Haemolysis is not produced on sheep blood agar. No growth on
Simmons’ citrate agar.
becoming more distinct with age. No water-soluble exopigment is formed. Growth is
stimulated by cysteine and thiamine or cysteine, thiamine, and pantothenic acid. In
nutrient broth produce moderate turbidity and deposit; in some strains a surface
ring and pellicle are formed. The optimal growth temperature is in the range 25-37 ºC.
No growth at 5 ºC. Grows well in 7.5% NaCl media. No growth in 10 or 15% NaCl
media. Aerobic. Haemolysis is not produced on sheep blood agar. No growth on
Simmons’ citrate agar.
Isolated from water and soil. Susceptible to tetracycline, erythromycin, oleandomycin,
novobiocin, methicillin, kanamycin, polymyxin, vancomycin, penicillin G, streptomycin,
chloramphenicol, and neomycin. Slightly resistant or resistant to lysozyme.
Resistant to gamma radiation; the level is uncertain, but it is in the region of 1 Mrad.
novobiocin, methicillin, kanamycin, polymyxin, vancomycin, penicillin G, streptomycin,
chloramphenicol, and neomycin. Slightly resistant or resistant to lysozyme.
Resistant to gamma radiation; the level is uncertain, but it is in the region of 1 Mrad.
Saprophytic. Reported to cause catheter-related bacteremia in patients with severe underlying diseases.
- Hans-Jurgen Busse, 2012. Family I. Micrococcaceae Pribham 1929, 361 AL emend. Stackebrandt, Rainey and Ward-Rainey 1997,
479 in: Bergey’s Manual of Systematic Bacteriology, second edition, Volume Five The Actinobacteria, Part A, Springer, 571-666. - Stackebrandt E., Koch C., Gvozdiak O. and Schumann P.: Taxonomic dissection of the genus Micrococcus: Kocuria gen. nov.,
Nesterenkonia gen. nov., Kytococcus gen. nov., Dermacoccus gen. nov., and Micrococcus Cohn 1872 gen. emend. Int. J. Syst.
Bacteriol., 1995, 45, 682-692. - Brooks B.W. and Murray R.G.E.: Nomenclature for "Micrococcus radiodurans" and other radiation-resistant cocci: Deinococcaceae
fam. nov. and Deinococcus gen. nov., including five species. Int. J. Syst. Bacteriol., 1981, 31, 353-360. - Rainey F.A., Nobre M.F., Schumann P., Stackebrandt E. and Da Costa M.S.: Phylogenetic diversity of the deinococci as determined
by 16S ribosomal DNA sequence comparison. Int. J. Syst. Bacteriol., 1997, 47, 510-514. - Maylraj S., Kroppenstedt R.M., Suresh K. and Saini H.S.: Kocuria himachalensis sp. nov., an actinobacterium isolated from the
Indian Himalayas. Int. J. Syst. Evol. Microbiol., 2006, 56, 1971-1975. - Kovacs G., Burghardt J., Pradella S., Schumann P., Stackebrandt E. and Marialigeti K.: Kocuria palustris sp. nov. and Kocuria
rhizophila sp. nov., isolated from rhizoplane of the narrow-leaved cattail (Typha angustifolia). Int. J. Syst. Bacteriol., 1999, 49, 167-
173.
Positive results for catalase, benzidine test, nitrate reduction (nitrite is not reduced),
acid production from L-arabinose, glucose, mannitol and D-xylose.
Can utilize as sole carbon source mannose and sorbitol.
Negative results for arginine dihydrolase, casein hydrolysis, coagulase (human and
rabbit), esculin hydrolysis, gelatin hydrolysis, beta-galactosidase, H2S production,
indole production, oxidase (rarely weakly positive), lecithinase, methyl red,
phenylalanine deaminase, phosphatase, Tween 80 hydrolysis, urease,
Voges-Proskauer test (sometimes weak), acid production from mannose, galactose,
glycerol, lactose, maltose, salicin, sorbitol, sucrose, turanose and trehalose.
No utilization of adonitol, L-arabinose, m-inositol and mannitol.
Variable results for starch hydrolysis.
acid production from L-arabinose, glucose, mannitol and D-xylose.
Can utilize as sole carbon source mannose and sorbitol.
Negative results for arginine dihydrolase, casein hydrolysis, coagulase (human and
rabbit), esculin hydrolysis, gelatin hydrolysis, beta-galactosidase, H2S production,
indole production, oxidase (rarely weakly positive), lecithinase, methyl red,
phenylalanine deaminase, phosphatase, Tween 80 hydrolysis, urease,
Voges-Proskauer test (sometimes weak), acid production from mannose, galactose,
glycerol, lactose, maltose, salicin, sorbitol, sucrose, turanose and trehalose.
No utilization of adonitol, L-arabinose, m-inositol and mannitol.
Variable results for starch hydrolysis.
(c) Costin Stoica

| Antibiogram |
| Encyclopedia |
| Culture media |
| Biochemical tests |
| Stainings |
| Images |
| Movies |
| Articles |
| Identification |
| Software |
| R E G N U M PROKARYOTAE |


| Back |